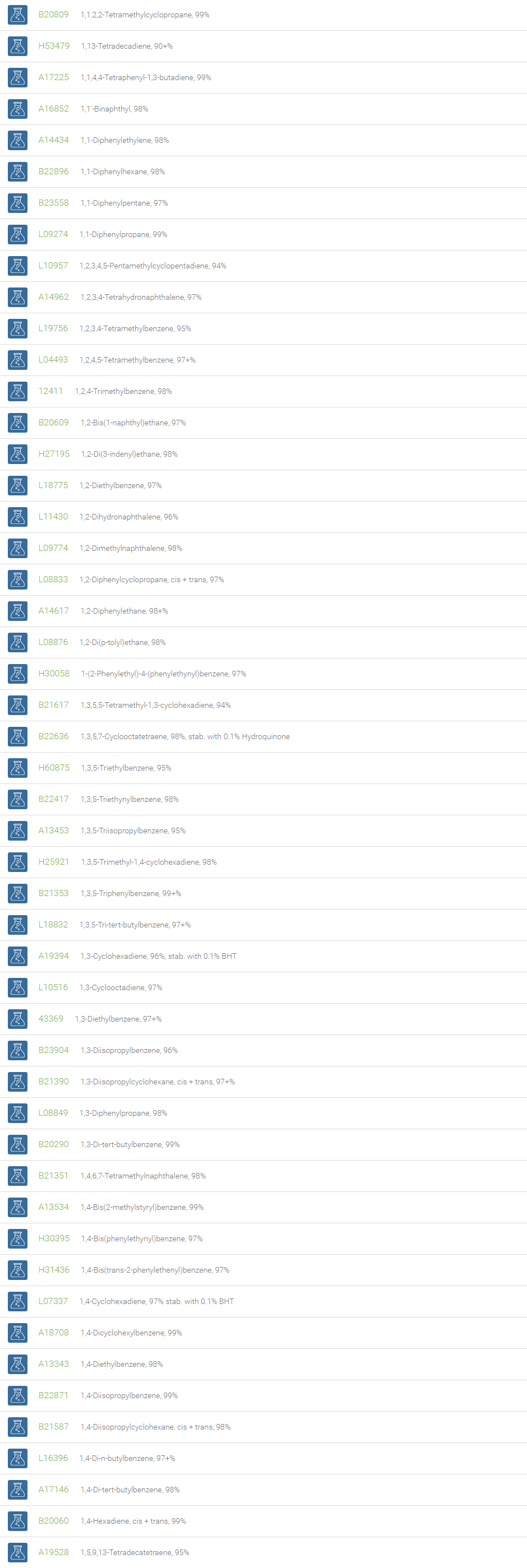
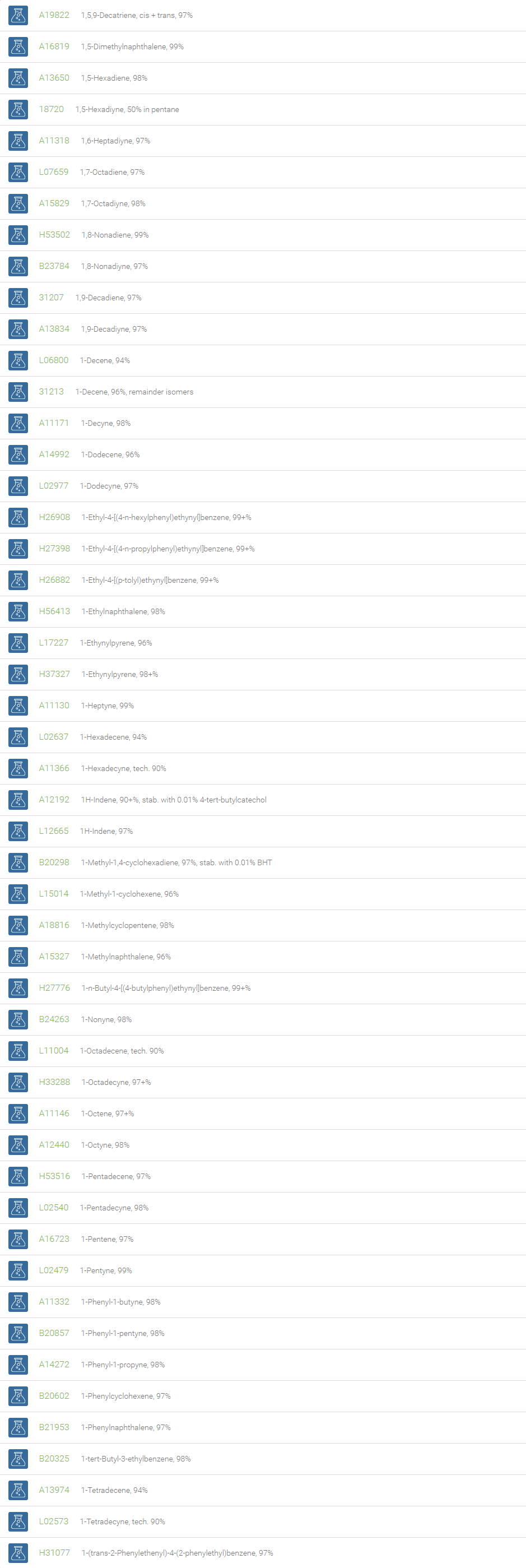
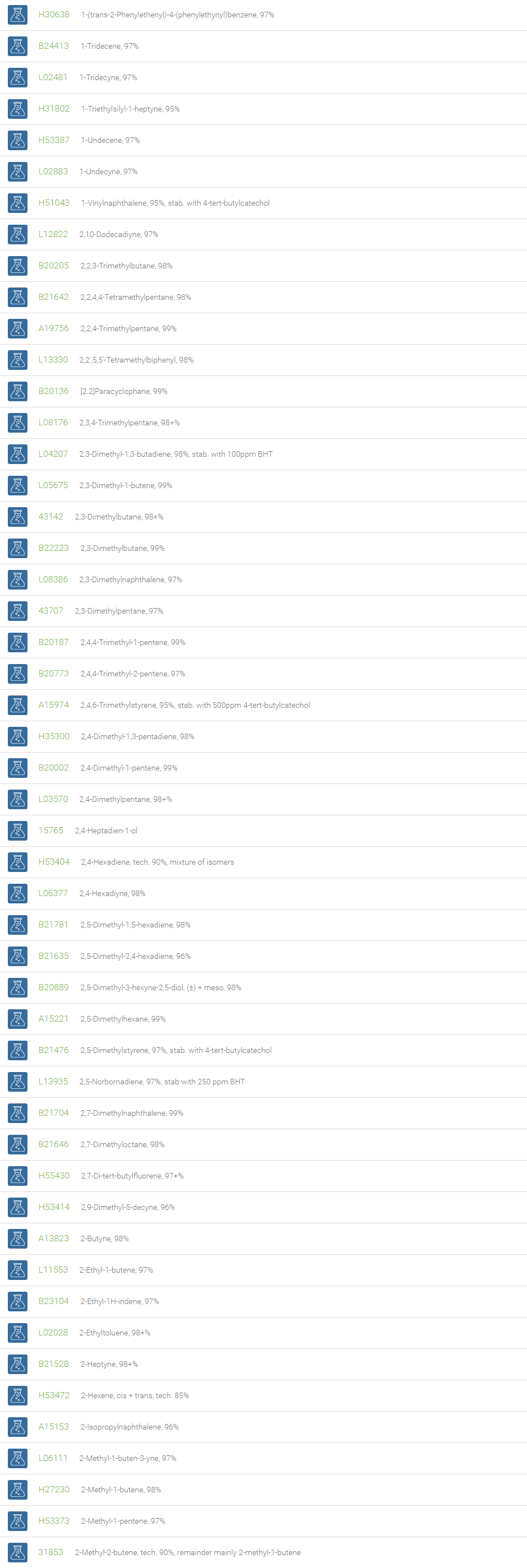
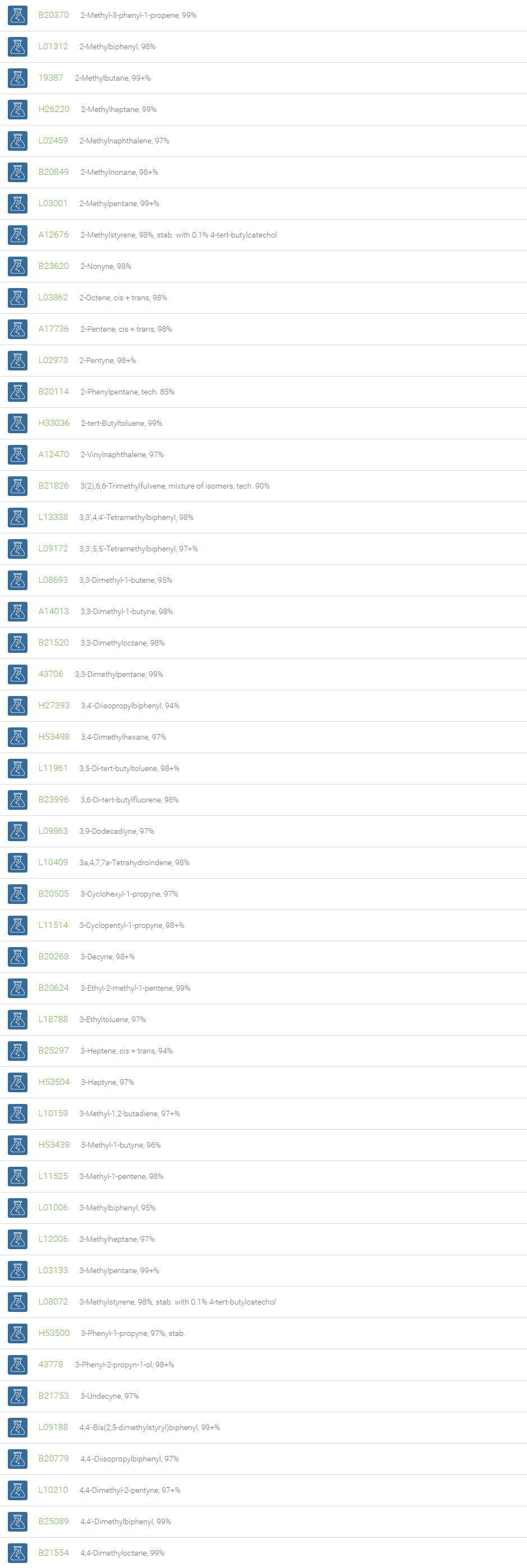
Alkanes, Alkenes, and Acetylenes
Alkanes, Alkenes, and Acetylenes
Alkanes, alkenes and alkynes are simple hydrocarbon chains with no functional groups. The simplest organic compounds are the alkanes. Alkanes have only single bonds between carbon atoms and are called saturated hydrocarbons. Alkenes have at least one carbon-carbon double bond. Alkynes have one or more carbon-carbon triple bonds. Alkenes and alkynes are called as unsaturated hydrocarbons. Alkanes have the general formula of CnH2n+2 where n is the number of carbon atoms. Alkenes have the general formula CnH2n. The general formula for alkynes is CnH2n-2. Acetylene is the simplest alkyne with the formula as C2H2.
Alkanes are non-polar compounds and insoluble in water. They have low boiling and melting points. In general alkanes & cycloalkanes have low reactivity & are used as solvents in organic chemistry. Natural gas, camping gas, lighter gas and much of gasoline are all alkanes. All the alkanes burn but they need a lot of air or oxygen to burn completely. Alkenes and alkynes are more reactive than alkanes. Unsaturated compounds (with double or triple bonds) form addition & cycloaddition compounds, where two substances combine to form a single substance.