Inorganic Acids

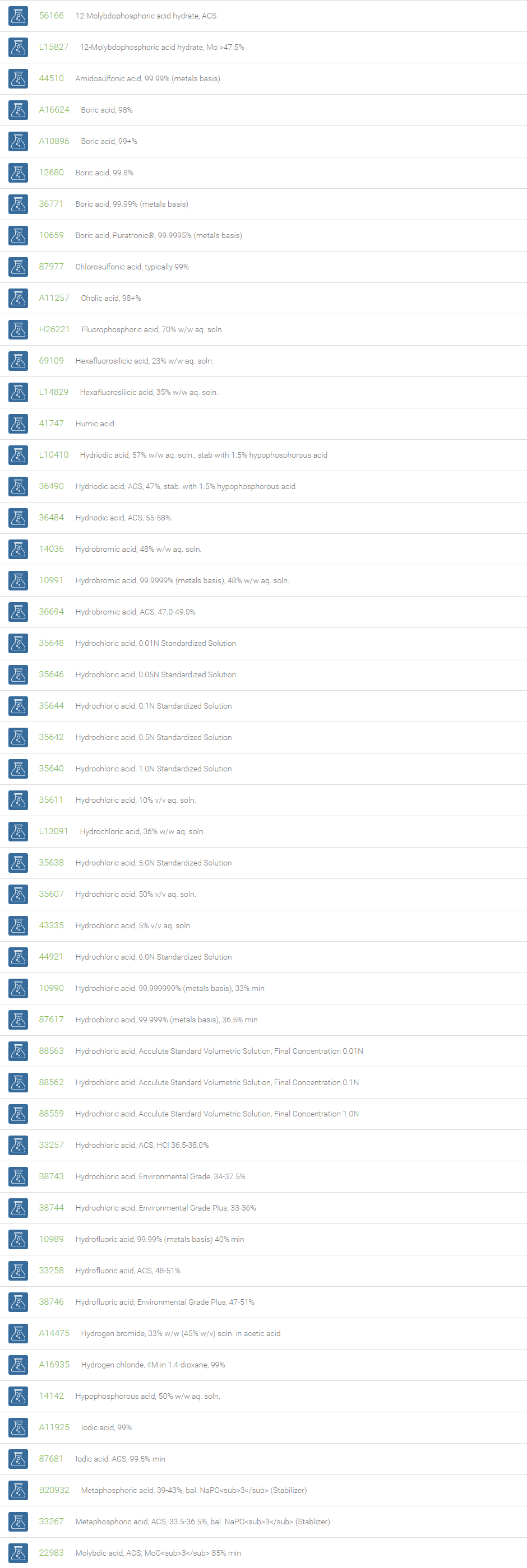
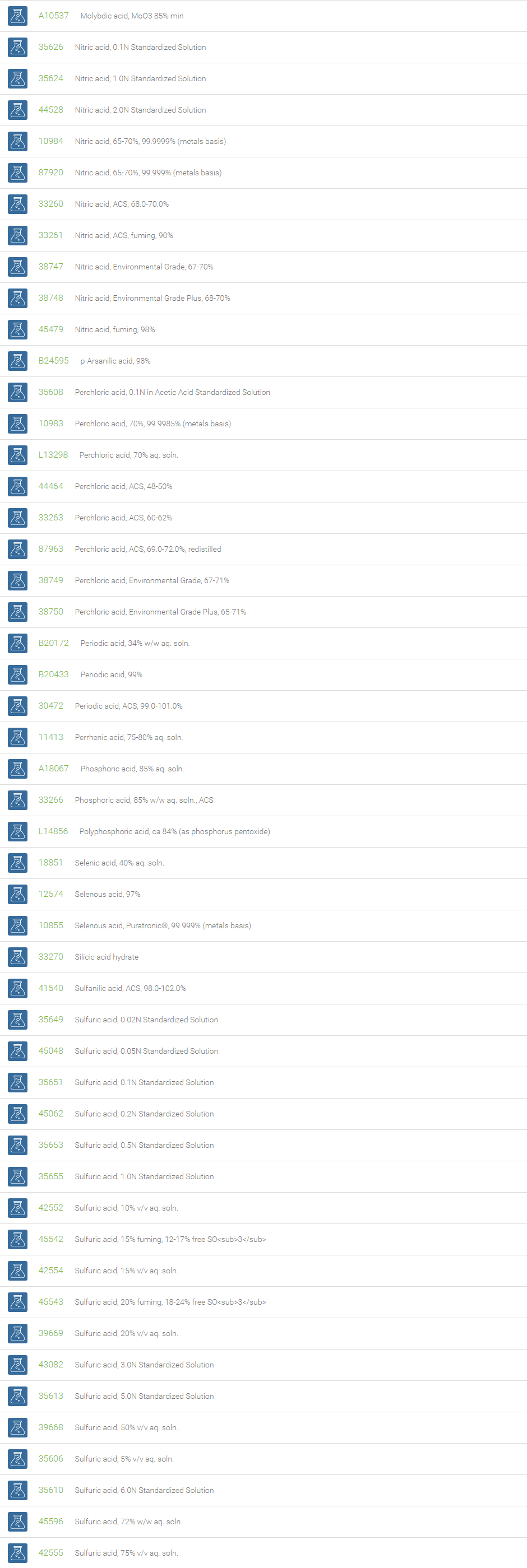
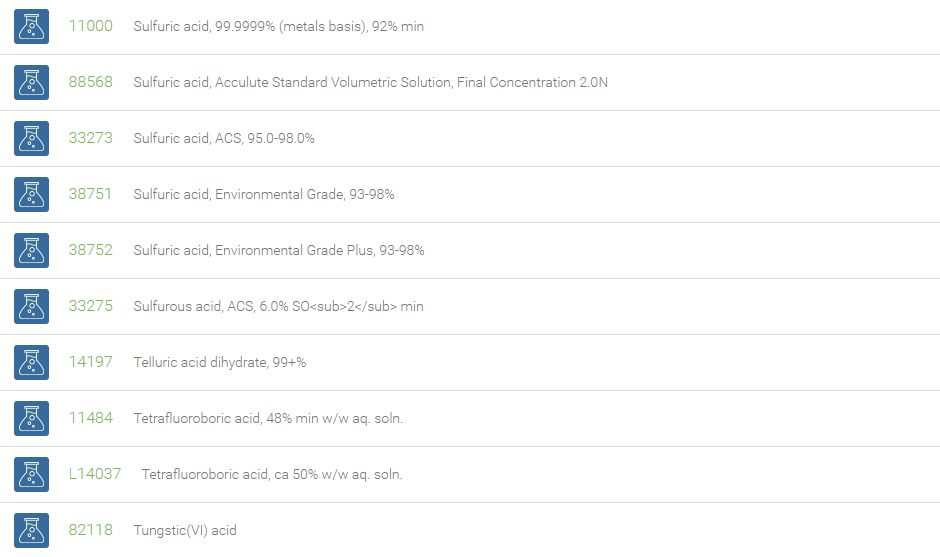
Inorganic Acids
Inorganic acids, which contain no carbon and are also called mineral acids, are acids derived from one or more inorganic compounds. These inorganic acids are either without oxygen or oxoacids. With reference to the number of hydrogen atoms they are either mono-, di-, or tribasic. Examples of acids without oxygen are: hydrochloric acid, hydrofluoric acid, which are monobasic and hydrogen sulphide, which is bibasic. Oxygen containing acids include, nitric acid (monobasic), sulfuric acid (dibasic) and orthophosphoric acid (tribasic). Inorganic acids when dissolved in water form hydrogen ions and the conjugate base ions.
Inorganic acids find applications in many sectors of the chemical industry as feedstocks for the synthesis of other chemicals. These acids are used in the manufacture of fertilizers, paints, dyes, plastics, synthetic textiles, and solvents. They are also used to make stock solutions for workup procedures in laboratories. In descaling, inorganic acids are used to remove deposits in boilers and reactors. Inorganic acids are excellent reagents for the elimination of water in organic synthesis. In esterification reactions they are used as catalysts and have been used in the hydrolysis of cellulose.